2 Which of the Following Represents a Known Ion
What peak represents the base peak. Oct 26 2016.

The Chemistry Of Gemstones Gemstones Minerals Geology
This 23 words question was answered by Jared M.
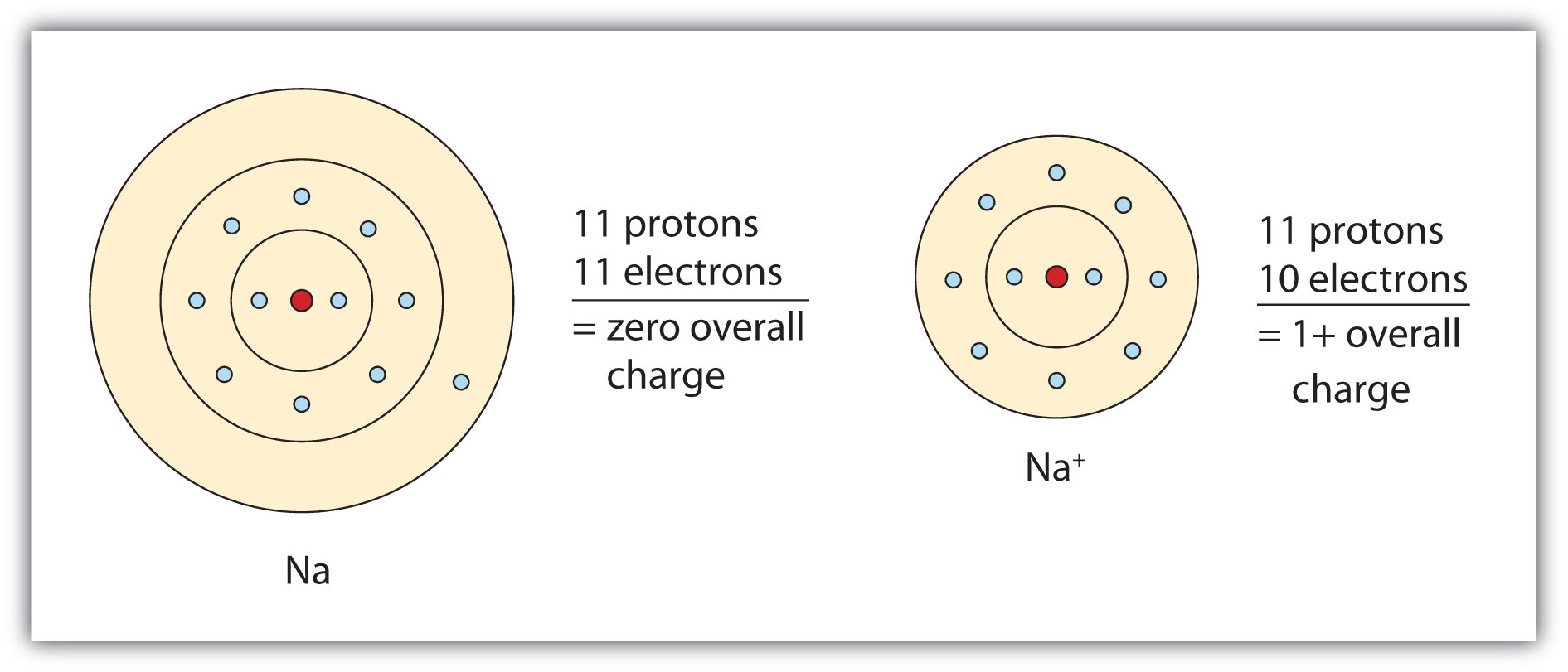
. Strong acid solutions could be harmful to human. Their electronic configuration is similar to that of neon Z 10 which is a noble gas element. In which of the following are the symbol and name for the ion given correctly.
___ is the heaviest noble gas. This is formed when an atom loses an electron to another atom. Chem 1110 - Chapter 2.
Identify the compound below which is an ionic compound. Because the ion has 16 protons and 18 electrons its net charge is 2. An ion is neutral and has no charge.
A CH 4 b H 2 O 2 c Na 2 CO 3 d NH 3 e SO 2. Which of the following is capable of forming a zwitter ion. It contains a Sub- threshold that changes in potential.
Bal2 is also known as Barium Iodide. Where l 0 corresponds to an s orbital l 1 means p orbital and so on. Determine the amount in moles of the following.
This particle without electrons would then have an ionic charge of 9. The formulas of the hydroxide ion the nitrate ion and the phosphate ion are represented respectively as a OH- NO3- PO43-. Which chemical formula represents the hydroxide ion.
Select the group of symbols that would correctly complete the following statements respectively. ___ is the transition metal that has 24 electrons as a 3 ion. ___ is the halogen in the third period.
View solution View more. View solution Among the following groups which represents the collection of isoelectronic species. 57 43 29 72 a.
CLASSES AND TRENDING CHAPTER. A particle with only 9 protons and no electrons would be an ion because one proton contains a 1 charge and one electron contains a -1 charge while neutrons have no charge. The three-carbon molecules broken down from six-carbon molecules of glucose during the first step in the process of nutrition in all organisms is called.
Skin eyes bones and lungs. Based on the given compounds the best answer is B. The first few atomic orbitals are 1s 2s and 2p.
Identify the compound formula that is INCORRECT. ___ is the alkaline earth metal that has 18. The dollar value to the right of each decision node represents the net cash flow at that point and the cash flows shown undert 3 4 and 5 represent the cash inflows if the project is pushed on to completion If Free Spirit Industries Inc.
This means that O 2- ion Na ion and Ne atom are isoelectronic species. Compound B is. Each orbital can hold 2 electrons maximum and there are 2l 1 of each type of orbital spdfg.
Its duration is directly proportional to the triggering event. 1215 g Mg. What peak represents M also known as the molecular ion or parent ion.
View solution The valency of a phosphate ion is. Which of the following represents an ion. Which of the following represents Plumbous ion.
Thus the symbol for the ion is. A Ca 3 PO 4 2 for calcium phosphate b NaNO 3 for sodium nitrate c K 2 CO3 for potassium carbonate d NH 4 SO 4 for. Thus each atom or ion of sulfur must contain 16 protons.
Any substance which yields a hydrogen ion when placed in a water solution is called an ____. Ca2 He H2 CO2 KCl. We are told that the ion also has 16 neutrons meaning the mass number of the ion is 16 16 32.
Based on the periodic table O is atomic number 8 which means it has 8 electrons. On StudySoup on 5312017. 2 CdF 2 D MgO MgS CaO E Al 2 O 3 In 2 O 3 Ga 2 S 3 12.
Those that come with opposite charge can be held together strongly within the compound. Elements A and B have atomic numbers 18 and 16. This is a type of iodide solution that is commonly used for medicinal purposes.
It occurs in varying grades short lived 2. An ionic bond should have a metal and a non-metal. This is a stable compound in dry places.
Some are depolarizing and some are hyper-polarizing. B H- NO2- P3-. In the following decision tree table 1 2 and 3 represent decision points also known as decision nodes or stages.
Propose a structure for the fragment ion at m2 57. Ca2 Carbon has 4 electrons and hydrogen has 1 electron in their outer most electron shells. How many hydrogen atoms can bond to one carbon atom.
1is a local electrical change in the membrane. Atoms Molecules and Ions Practice Quiz 1. The following is the mass spectrum of 2-methylbutane.
Both Na ion and O 2-ion are known as isoelectronic species as they have same number of electrons ie. Acetate C2O4- oxalate NH4.

This Is How The Ionic Bond Forms In Sodium Oxide Na2o Ionic Bonding Bond Ionic

Ionic Compound In 2022 Ionic Compound Ionic Oxygen

Organic Chemistry Educational Infographics Chemical Reactions Organic Chemistry Introduction To Organic Chemistry Organic Chemistry Reactions

Ncert Solutions For Class 11th Chemistry Chapter 4 Chemical Bonding And Molecular Structure 11th Chemistry Chemical Bond Chemistry

Chip Carving Chip Carving Carving Engraved Wood

Ionic Bonding Worksheet Teaching Resources Ionic Bonding Ionic And Covalent Bonds Bond

Jasmine On Instagram Tip Use Symbols And Abbreviations To Keep Up With Lectures My Favorite Is Using What Looks Like An Exponen Lecture Exponents Education

Classify These Structures By The Hybridization Of The Central Atom In 2022 Vsepr Theory Atom Central

Always Still Love You Double Hearts Necklace Gift For Daughter Message Card Favo Jewelry Standa Valentine Gift For Daughter Double Heart Necklace Gift Necklace

Coordination Chemistry Notes Chemistry Notes Transition Metal Chemistry

Pin On Education Studying Academics

Molecules And Compounds Overview Atomic Structure Article Khan Academy

Ktm Ion Concept Motorcycle For Urban Environment Tuvie Concept Motorcycles Electric Motorcycle Ktm

Element Charges Chart How To Know The Charge Of An Atom Chemical Chart How To Know Chemistry Periodic Table

Sodium Chloride Ion Salt Middle School Chemistry College Chemistry Chemistry

G Shock Mud Resistant Resin Band Men S Watch Black Gemorie In 2021 G Shock Watches Casio G Shock Mudman Sport Watches

3rd Grade Math Fractions In 2021 3rd Grade Math Fractions Math



Comments
Post a Comment